Medical records hold crucial patient information. These records are also a part of clinical trials, based on which new drugs and treatment approvals are given. Ensuring the confidentiality of such records is also regulatory compliance. Failure to meet them can result in lawsuits and millions lost in penalties. Keeping patient information safe and private is, therefore, one of the biggest concerns. Redaction of medical records is necessary to protect sensitive patient information. This article is about the Redaction of medical records and its importance for healthcare institutions.
Table of Contents
What is the Redaction of Medical Records?
Redaction of medical records is the process of removing sensitive information from any document. Healthcare institutions use redaction to ensure the information they share internally or externally does not compromise anyone’s privacy or security. Redaction also makes it easy to publish documents that contain sensitive information.
Almost 15 million Americans become victims of identity theft each year. Data Redaction is thus essential to protect an individual’s personal information and keep their identity private. It holds much more importance in sensitive sectors like medicine and healthcare. Data redaction is sometimes confused with data anonymization. However, information is hidden in data anonymization, whereas information is fully deleted in data redaction.
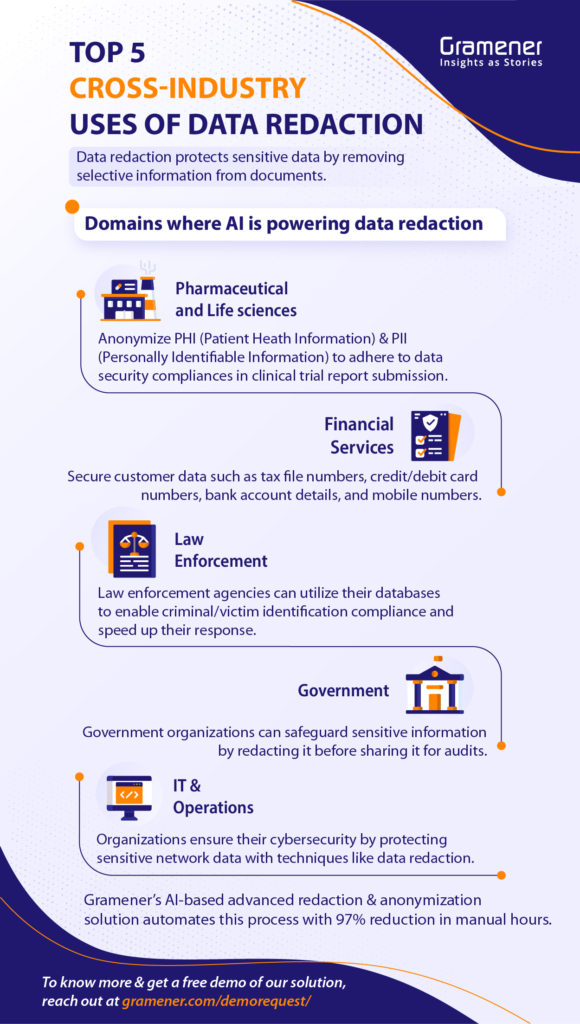
How Does Redacting Work?
Redaction of medical records is a simple process that requires only three steps:
- Scanning of documents to identify Personally Identifiable Information (PII) for the redaction process
- Removing all Personally Identifiable Information (PII)
- Storing of redacted files for future use
Medical Document Redaction Best Practices
Here are some best practices to keep in mind regarding the Redaction of medical records-
Avoid Redacting the Original Copy
It is always best to save an additional copy of the document you want to redact. If you accidentally redact something you did not mean to, you’ll lose it forever. So, archive a copy that you’re editing to ensure that it is safe. Then, go through each page of a separate copy to redact private information.
Redact Files in PDF Format
Redacting files in a PDF format will ensure that the information does not fall into the wrong hands. If unscrupulous individuals try to convert the documents into other formats like word, they will lose all the sensitive information. Redaction of medical records in a word document does not offer protection for sensitive data.
Avoid Redacting Digital Documents Directly
When redacting manually, it is best to print the scanned or photocopied document first. You can use a black marker to hide the information. The last step in the redaction of medical records here will be to scan the paper again and convert it into PDF format.
Take Care of the Metadata
Ensure that you remove all the attributes at the time of the redaction of medical records. If not, unauthorized persons may still be able to view sensitive information through the document’s metadata. It is best to eliminate the entire metadata to prevent chances of someone gaining unlawful access to private information.
Read More: Find out the best data anonymization tools to redact and mask information in any document.
Importance of Data Redaction in Pharma and Life Sciences?
Redaction of medical records is essential in the pharma and life sciences industries because of the strict regulations around patient data. You may be working on the drug discovery process and clinical trials. You must know how to redact sensitive data, so you do not inadvertently expose it.
Several laws mandate the need for redaction in the drug discovery process and clinical trials. For example, HIPAA requires that certain data types be redacted for privacy before sharing it with the public or third parties. It also requires organizations to have a process to ensure data gets redacted for privacy before going out into the public domain.
Another reason redacting medical records is essential is because it helps protect patients’ privacy rights. When someone’s private information gets exposed without their consent, they can sue your company for violating their privacy rights under applicable laws. These lawsuits can cost millions of dollars in damages and generate bad press.
What Data Should Be Redacted from Medical Records?
Here are the different information types eliminated during the redaction of medical records.
Potentially Harmful Information
Redaction of medical records can help healthcare providers and the organizations they work for and protect patients and their families. Several regulations such as GDPR and EMA 0070 require organizations to ensure data privacy with transparency and public disclosure, making it imperative to protect patient privacy and ensure confidentiality by redacting personal information from medical records.
This can include names, addresses, phone numbers, social security numbers, birthdates, and anything else that can help identify an individual. Nefarious individuals can use these data points to harm an individual physically or mentally. It is thus essential to safeguard this data.
Third-party Information
It is critical to protect third-party information in medical documents as they can be shared with someone without the right to access it. It could lead to identity theft or other fraud.
You might also have your confidential information used against you by people who do not have your best interests in mind. Similarly, a patient’s relative may have to reveal more information to doctors in the absence of the patient. Protecting their interests is also essential. Redacting medical records to protect everyone’s interests is thus necessary.
Meeting HIPAA Norms
Health Insurance Portability and Accountability Act (HIPAA) mandates healthcare providers to protect the personal health information of patients. It also prohibits such institutions from publishing patient records without consent. Redaction of medical records, under HIPAA guidelines, involves concealing individual identity details and specific information that can identify a person.
Patient Privacy and Redaction in the Medical Records
It is the responsibility of healthcare organizations to ensure that they are compliant with HIPAA regulations. This includes doctors’ offices, hospitals, insurance companies, medical billing services, medical equipment suppliers, and other health care providers.
Patients’ Rights Under HIPAA
The HIPAA guidelines include several provisions that protect an individual’s rights as a patient. These include the right to:
- View and copy health records
- Request restrictions on how to use or disclose health information
- Request confidential communications of health information
- The right to have a person of choice accompany for an inspection, review, or copying of medical records
Information Protected Under HIPAA
Redacting medical records protects the following types of information under HIPAA –
- Information added by healthcare providers in medical records
- Conversations between doctors and nurses about patients’ treatments
- Patient information in the health insurance provider’s logs
- Patients’ billing details at their clinic
Who is Eligible for HIPAA, and How Do they Comply?
Entities covered under HIPAA redaction requirements include healthcare providers, payment and operations units, business associates, and other related agencies. They need to ensure compliance by creating privacy and security policies, naming HIPAA officers, and conducting regular audits.
PHI Under HIPAA Compliance: Overview
Protected Health Information (PHI) Protection applies to a person’s medical history or treatment. This can include a person’s name, address, social security number, genetic information, and even photo. PHI gets protection by law under HIPAA redaction requirements, which means that no one can disclose it without explicit consent from the individual.
For example, healthcare institutions conducting clinical trials cannot share the data with pharma and drug companies if they do not receive consent from individuals.
How to Protect Protected Health Information (PHI)?
The U.S Department of Health and Human Services introduced HIPAA in 1996 to safeguard the sensitive information of patients. It got further bolstered with the addition of the HITECH act in 2009. These acts together offer comprehensive security and privacy for PHI.
Healthcare providers and related business associates dealing with PHI need to comply with HIPAA laws. The PHI data under HIPAA redaction requirements can include everything from physical and digital data to spoken words.
The privacy breach of PHI can result in severe consequences. It is a criminal offense that can also invite penalties. American Medical Association lists that a PHI violation can invite a penalty of up to $50,000. Repeat offenses can incur fines of up to USD 25,000 in a year.
Bottomline
With the increasing emphasis on patient privacy and confidentiality, it has become imperative for healthcare institutions to safeguard patient data. It’s best to prevent the leakage of sensitive information such as addresses, phone numbers, and social security numbers. There might be instances when miscreants can identify patients if appropriate compliances and redaction best practices are not followed. Data redaction is the best option to avoid such situations.
Gramener is a leading provider of cutting-edge data redaction solutions for the healthcare industry. We are helping global healthcare majors protect their patients’ privacy and meet HIPAA compliance.
Contact us for custom built low code data and AI solutions for your business challenges and check out pharma and life sciences AI solutions built for our clients, including Fortune 500 companies. Book a free demo right now.


What are the STEPS to REDACT INFORMATION.
Only need STEPS.